Publications
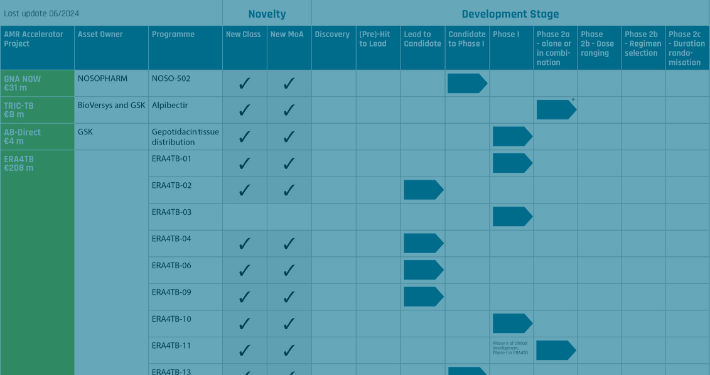
The AMR Accelerator calls to action:
European capacity for antibiotic R&D requires long-term funding
Fernow J, Olliver M, Couet W, et al. The AMR Accelerator: from individual organizations to efficient antibiotic development partnerships. Nature Reviews Drug Discovery 2024. Published online September 23, 2024. doi:10.1038/d41573-024-00138-9